Difference Between Magnesium and Magnesium Oxide
Magnesium and magnesium oxide are two closely related substances that are often easily confused with each other. However, they come with several distinctions.
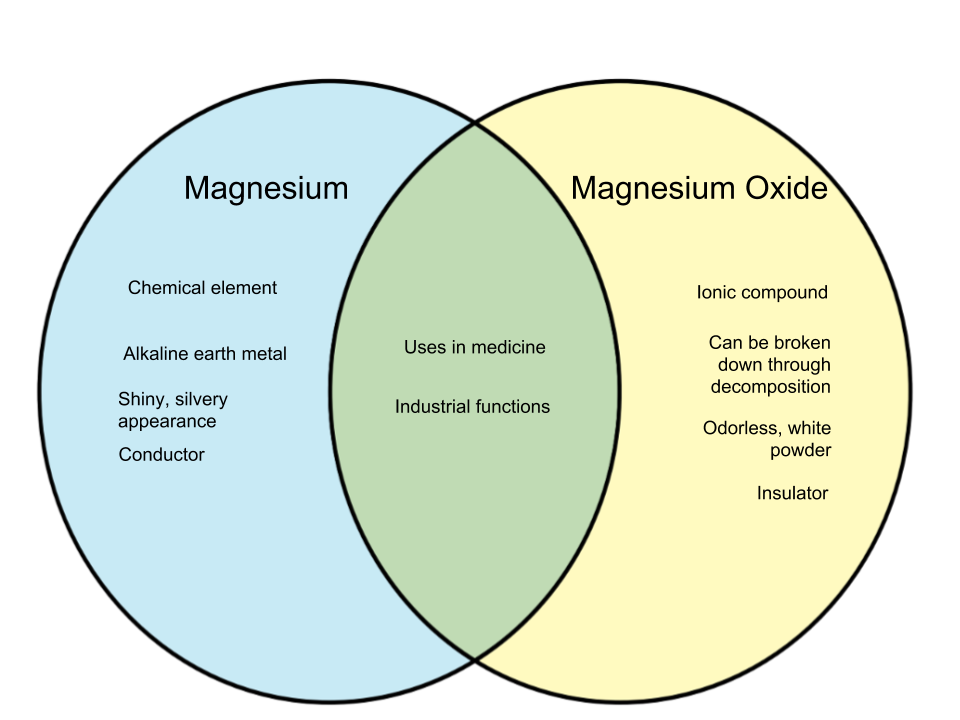
Magnesium is an element - a base substance that can be found listed in the periodic table of elements. It is an alkaline earth metal that has the capability to form different compounds. Magnesium possesses a shiny, silvery appearance and manifests itself in its solid state at room temperature. The element can be found in mineral deposits as well as seawater. It is also present in organic foods especially those rich in fiber. Magnesium can also be found in the human body - it plays a role in strengthening bones and helps circulate blood.
The element has a vast amount of uses - it has applications in metal, aircraft and automotive design, as well as medicine. Magnesium is applied in the treatment of several diseases and conditions, and also aids in controlling anxiety and treating cardiovascular diseases.
Magnesium oxide is an ionic compound that is formed when magnesium is combined with oxygen. It can be broken down using decomposition and is usually in the form of an odorless white powder. Magnesium oxide serves an an insulator but is also used mainly as a magnesium supplement, It is actually one of the most affordable and popular choices as supplement. As medication, it is consumed as a laxative, but is also believed to help relieve headaches, regulate blood pressure, and maintain blood sugar levels.
| Magnesium | Magnesium Oxide | |
|---|---|---|
| Classification | Element; alkaline earth metal | Ionic compound |
| Symbol | Mg | MgO |
| Appearance | Shiny, silvery gray | White powder |
| Melting point | 650 °C | 2800 °C |
| Source | Mineral deposits, seawater, organic food, foods rich in fiber | Combining magnesium and oxygen |
| Molar mass | 24 g per mol | 4.304 g per mol |
| Conductor or insulator | Conductor | Insulator |