Difference Between Osmosis and Diffusion
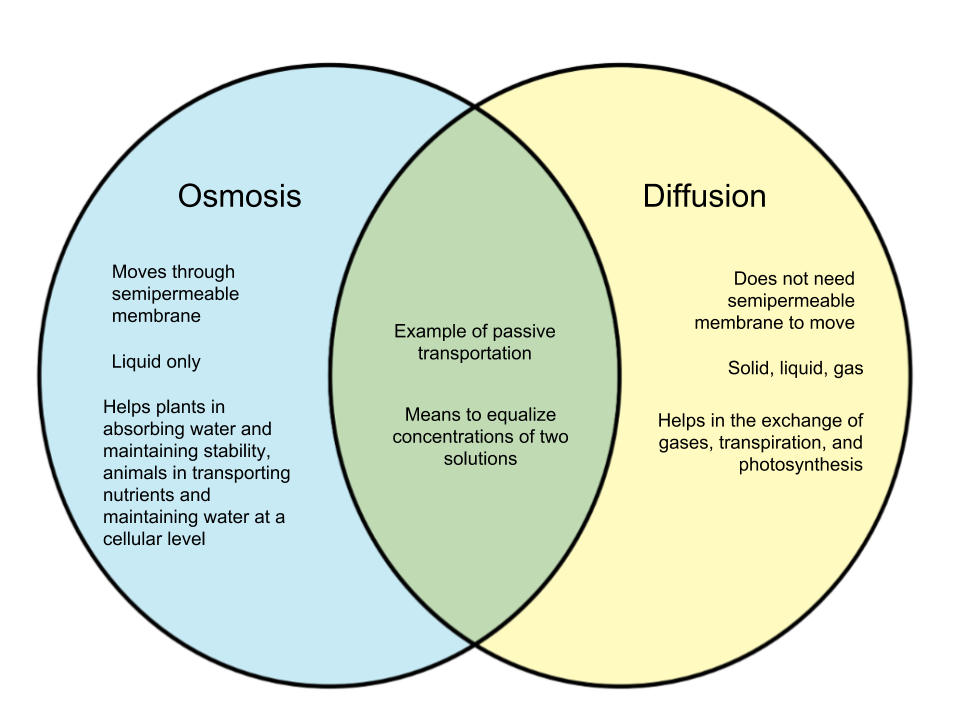
Both osmosis and diffusion are examples of passive transportation wherein energy does not need to be exerted to produce an effect. Both are also a means to make the concentration of two solutions equal. Here, we will discuss how osmosis and diffusion differ from each other.
Osmosis[edit]
Osmosis is the process where liquid moves from a higher region of concentration to a lower region of concentration through a semipermeable membrane. Usually, the solvent or liquid involved in this process is water. Osmosis is an essential process that helps animals transport nutrients and maintain water on a cellular level, as well as help plants absorb water from the soil.
Diffusion[edit]
Diffusion is the movement of particles from a higher region to a lower region of concentration. These particles can be solid, liquid, or gas. Unlike osmosis, particles do not move through a semipermeable membrane. It is a faster process than osmosis, which is quite slow. Diffusion is important because it allows processes like exchange of gases when animals respire or assists in transpiration and photosynthesis for plants.
| Header text | Osmosis | Diffusion |
|---|---|---|
| Definition | The process where liquid moves from a higher region of concentration to a lower region of concentration through a semipermeable membrane | The movement of particles from a higher region to a lower region of concentration |
| Medium | Liquid | Solid, liquid, or gas |
| Semipermeable membrane | Through the membrane | Does not need the membrane |
| Affecting factors | Temperature, diffusion distance, concentration gradient | Temperature, concentration gradient, pressure, molecular weight |
| Importance | Helps animals in transporting nutrients and maintaining water on cellular levels; provides support for plants and absorbing water from the soil | Helps animals’ respiration and transpiration and photosynthesis for plants |
| Examples | Soaking something in water, root pressure, wrinkled fingers from excessive exposure to water | Spraying perfume or freshener, cigarette smoke scattering in the air, dunking a tea bag in water |